diCELLa eCRF clinical trials
diCELLa eCRF clinical trials is a web-based application that enables the establishment and management of CRFs in non-commercial clinical trials.
The application is designed for both the sponsor/CPO and the research group. Data in diCELLa eCRF clinical trials are protected from unauthorized changes by using blockchain technology to ensure non-repudiation.
With our app you easily create and manage CRF!
The application is designed for both the sponsor/CPO and the research group. Data in diCELLa eCRF clinical trials are protected from unauthorized changes by using blockchain technology to ensure non-repudiation.
With our app you easily create and manage CRF!

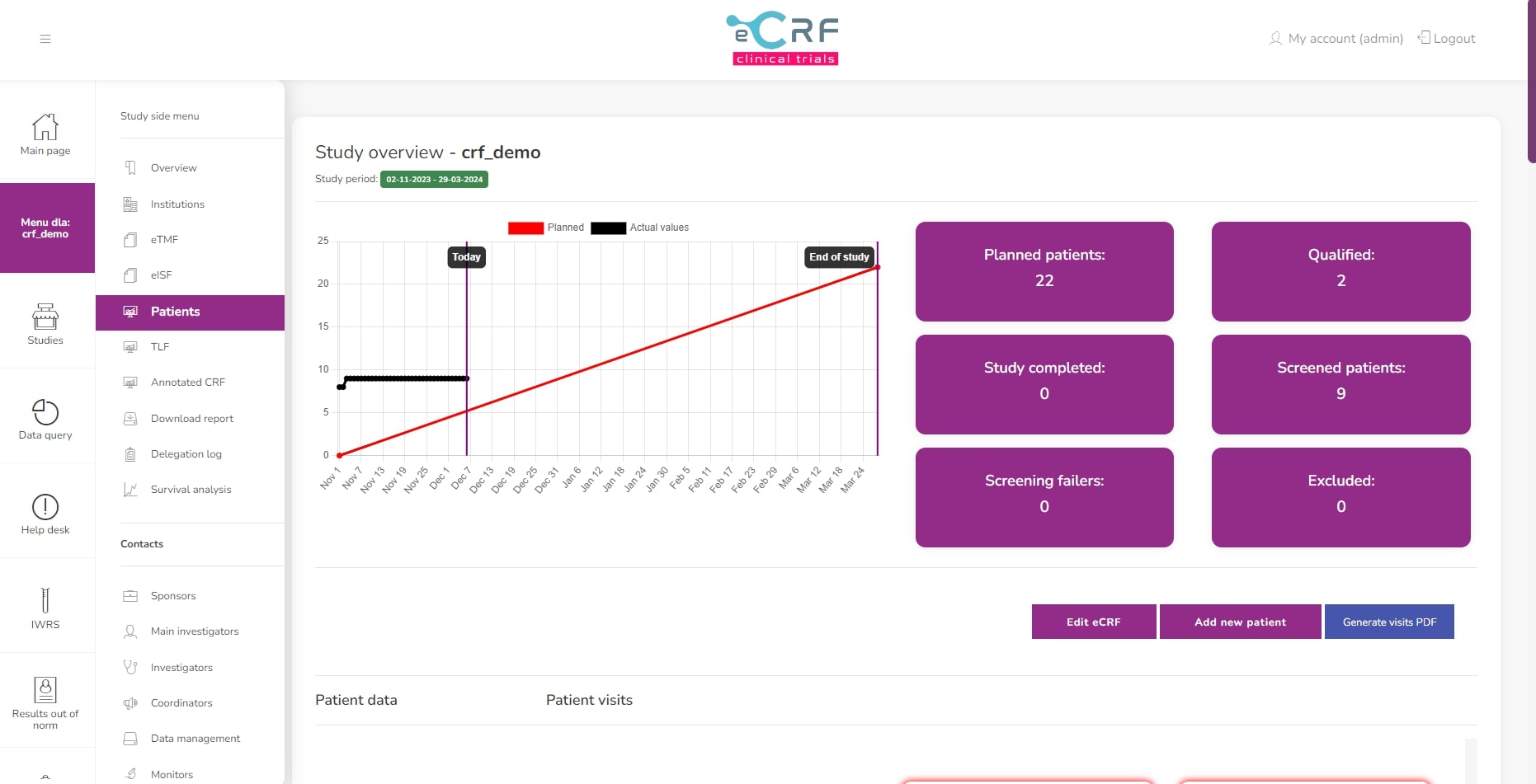
Patients statistics

Patients list
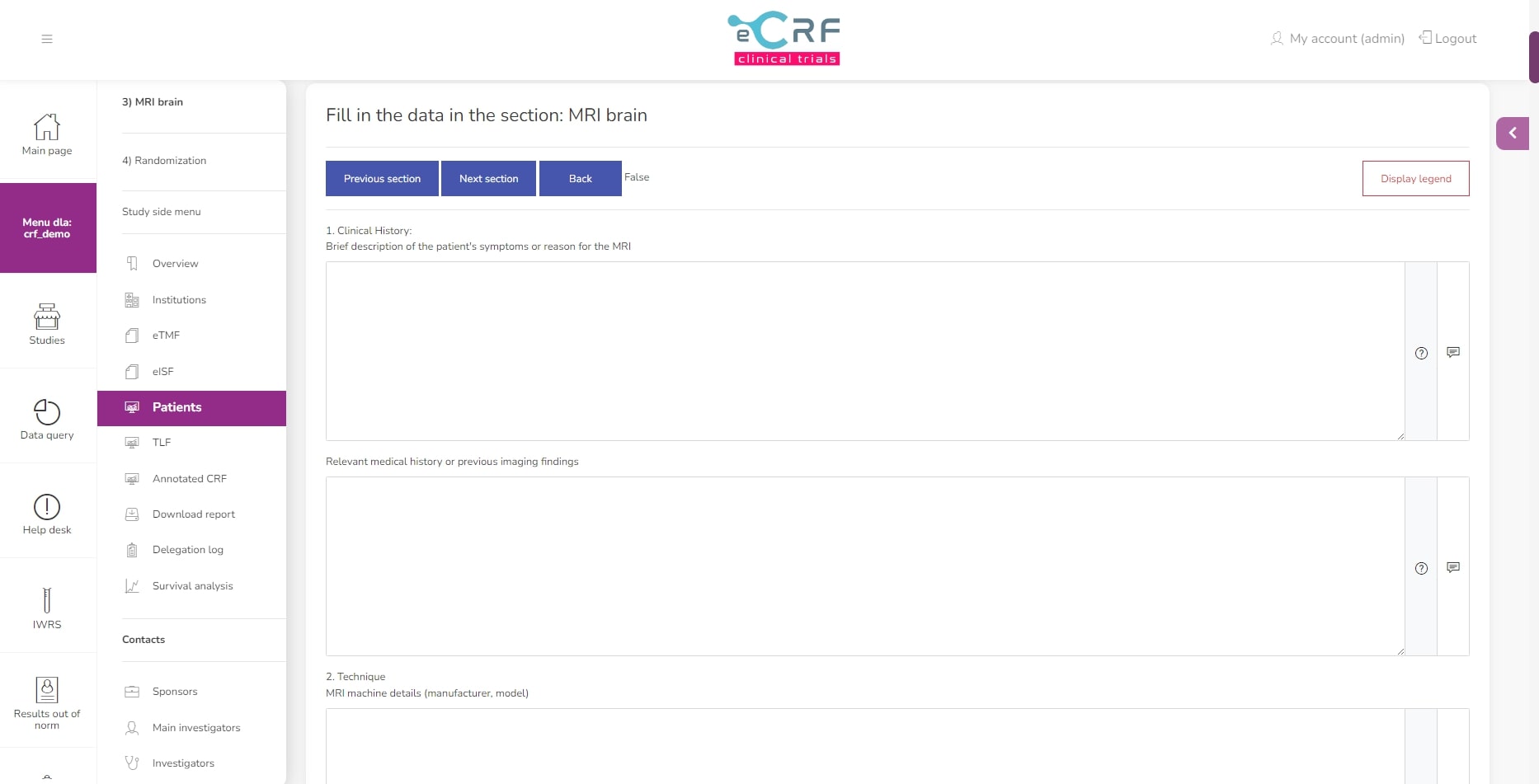
Questions in the section
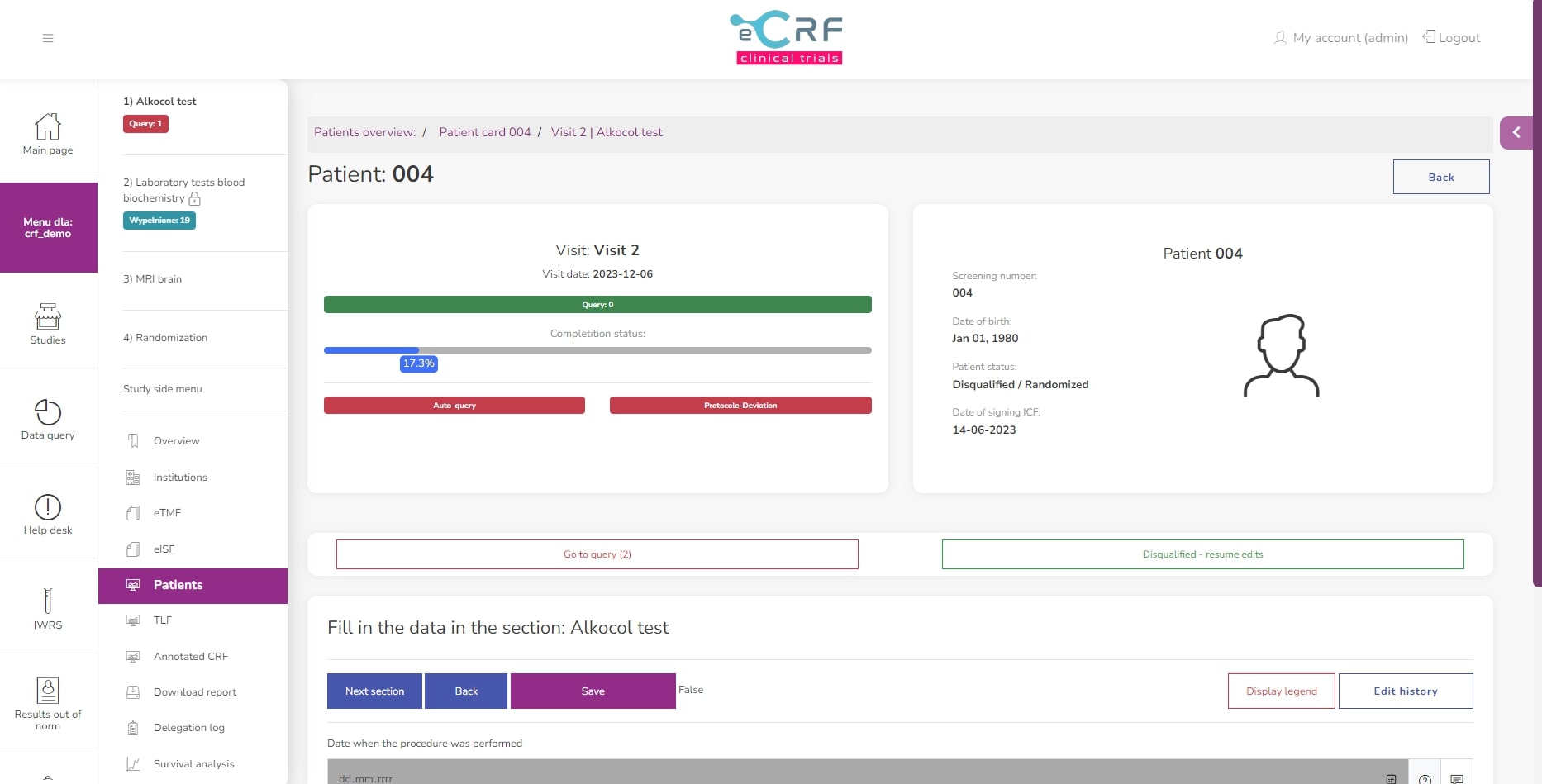
Section view
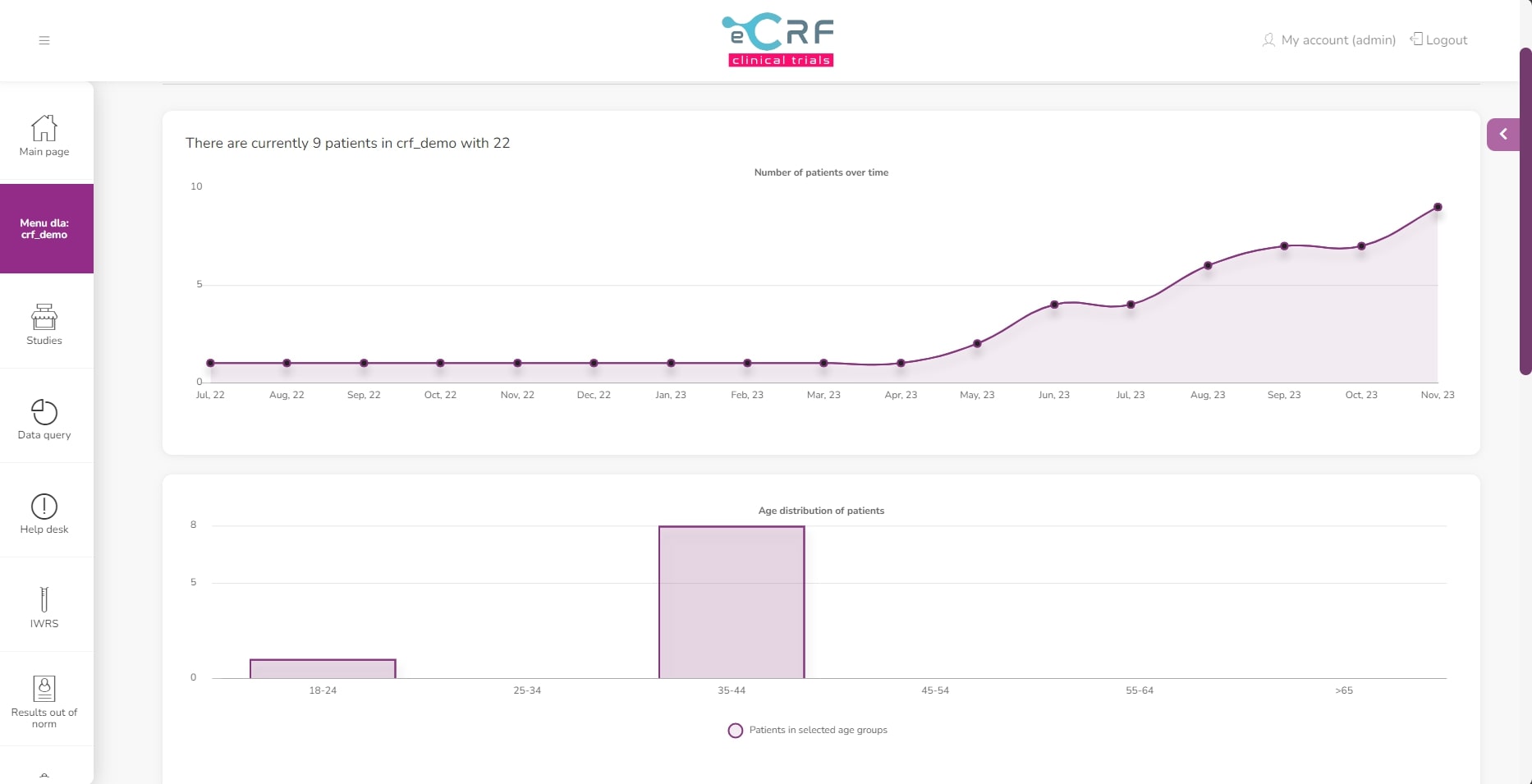
View of general study statistics

Query view
diCELLa eCRF clinical trials
Application for creating and managing CRFs
in non-commercial clinical trials
Application for creating and managing CRFs
in non-commercial clinical trials
The application is designed for both the Study Sponsor/CRO and the research team. As a study sponsor (and/or CRO), you will quickly create a CRF for a new clinical trial, be able to keep track of the progress of the clinical trial and make monitoring visits. As a research team, you will be able to easily complete CRFs of study participants.
The data in diCELLa CRF Clinical Trials are protected from unwanted changes through BLOCKCHAIN technology, ensuring authenticity and non-repudiation.
diCELLa eCRF clinical trials is dedicated to non-commercial early-phase clinical trials with strong investigator participation at the trial design stage.
The eCRF clinical trials is compatible with tissue donor qualification under the Act of July 1, 2005 on the Collection, Storage and Transplantation of Cells, Tissues and Organs (along with implementing acts), facilitating work with autologous medicinal products.
Research team: diCELLa eCRF clinical trials reflects the structure of the research team, monitor and sponsor in a clinical trial.
Sponsor/CRO: eCRF clinical trials will allow CRFs to prepare for new clinical trials and easily manage ongoing trials.
diCELLa eCRF clinical trials in any eCRF component can easily print periodic reports on donors and collected tissues/cells.
The data in diCELLa CRF Clinical Trials are protected from unwanted changes through BLOCKCHAIN technology, ensuring authenticity and non-repudiation.
diCELLa eCRF clinical trials is dedicated to non-commercial early-phase clinical trials with strong investigator participation at the trial design stage.
The eCRF clinical trials is compatible with tissue donor qualification under the Act of July 1, 2005 on the Collection, Storage and Transplantation of Cells, Tissues and Organs (along with implementing acts), facilitating work with autologous medicinal products.
Research team: diCELLa eCRF clinical trials reflects the structure of the research team, monitor and sponsor in a clinical trial.
Sponsor/CRO: eCRF clinical trials will allow CRFs to prepare for new clinical trials and easily manage ongoing trials.
diCELLa eCRF clinical trials in any eCRF component can easily print periodic reports on donors and collected tissues/cells.
